Transarterial chemoembolization (TACE) is a commonly used method for treating early-stage liver cancer.
The GSEP based on medical-grade absorbable gelatin achieves precise embolization through controlling the particle size and adopting a non-irradiation-resistant design.
- Safe and reliable: It has excellent biocompatibility, is fully biodegradable, and leaves no long-term foreign matter residue.
- Operational flexibility: Different particle sizes can be selected, making injection easy, and the clear radioactive imaging effect facilitates the achievement of precise embolization.
- Economically efficient: Mature technology, high cost-effectiveness, wide applicability.
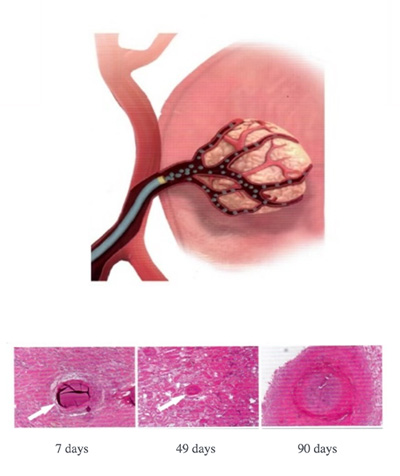
Reliable targeted embolization technique:
The appropriate model can be selected based on the location of the embolus and the size of the blood vessel.
Under the guidance of digital subtraction angiography (DSA), it can precisely reach the target area with extremely small deviations.
High compressibility:
The porous sponge structure can achieve a compression deformation of over 50%.
After being removed from the catheter, it can quickly return to its original state, thereby achieving a tight embolization effect in the distal blood vessel.
Low friction coefficient, with very little aggregation:
It is manufactured through a refined collagen processing technology, which can prevent aggregation from occurring within the catheter and in the human blood vessels.
Intracellular degradation and absorption characteristics:
After implantation, it will cause irreversible ischemic necrosis at the target site.
It gradually decomposes over a period of approximately 90 days.